
Packaging designers may need to adjust graphics on food products to make room for new information boxes proposed by the U.S. Food and Drug Administration (FDA).
Food and beverage brands and other industry partners gave mixed reactions to the agency’s nutrition information box design Tuesday as they began digesting long-awaited proposed front-of-package labeling rules. The essential, compact, standardized boxes are intended to complement the FDA’s existing Nutrition Facts label, which was last updated in 2016.
While some groups have rejected the FDA’s methodology for full evolution, other sources have pointed out that the proposed format still appears softer than other countries’ approaches.
Natalie Rainer, a partner at K&L Gates, noted that the FDA’s proposal appears to reflect a neutral, consistent aesthetic for nutrition labels and may be less alarming than incorporating traffic light colors or internationally used stop sign graphics. Still, “I think it’s going to take up too much essential real estate that businesses didn’t have to sacrifice before,” she said.
“It’s a pretty big change in the way the FDA handles nutrition labeling,” Rainer said, nodding to the agency’s move to assign low, medium and high values to various nutrients. “The nutrient content regulations to date have given companies guardrails on how to make favorable claims about their products. This has led them to make some sort of negative statement.”
Christy Lebor, group director of strategy at Smashbrand, a CPG packaging design and branding agency, said of the aesthetic impact:“I am concerned.” Especially when you consider how it would work for smaller packaged items like single packs. A box of gum or Jell-O.
“Large, prominent callouts, often referred to in the industry as ‘offenders’ or ‘yum yum,’ detract from the visual appeal of packaging, regardless of the health status of the product. Lebor said in a written statement:

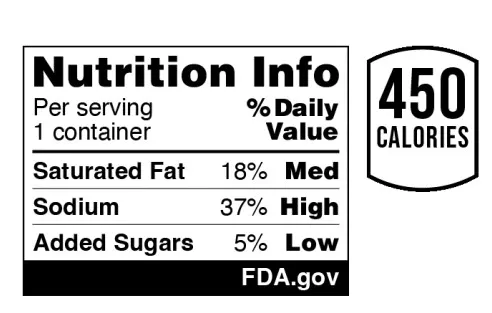
Provided by the U.S. Food and Drug Administration
Suggested Specifications for Nutrition Facts Box
- “Low” ranges from 5% or less of daily value; 6% to 19% DV for “Med”; 20% DV or higher for “high”
- The box will be placed somewhere in the top third of the main display panel (the FDA does not specify the exact location).
- The FDA recommends, but does not specify, a single type of style that is easy to read. This example uses Helvetica, as does the nutrition facts label.
- The FDA suggests a minimum type size of 8 points and provides other specifications for the basic size required for the carton relative to the total packaging area.
- The FDA has also proposed a more basic “intermediate” version of the information box that can be used on smaller packages (packages less than 40 square inches that can be labeled).
The FDA’s approach has drawn mixed reactions.
Federal action on front-of-package labeling has been a long time coming.
Nonetheless, the proposed rule “came out in the 11th hour of the Biden administration,” Sam commented. jokelHe is a partner at Alston & Bird, and it remains to be seen whether the incoming Trump administration may change course.
The FDA has been involved in front-of-package nutrition labeling issues for nearly 20 years. The Center for Science in the Public Interest (CSPI), a nonprofit consumer advocacy group, said it petitioned the FDA for front-of-package nutrition labeling in 2006 and again in 2022.
The FDA said it tested several design options, some of which included a magnifying glass icon or used traffic light-inspired colors (red, yellow, green), but is moving forward with a text-only black-and-white design. The rule states that although some information suggests that color coding with text may improve comprehension, the FDA’s own research has not shown a statistically significant benefit. The boxes do not list calorie counts, but the agency said brands can choose to display that information simultaneously.
The Consumer Brands Association, which represents dozens of CPGs, criticized the FDA’s approach.
“FDA’s proposed front-of-pack nutrition labeling rule appears to have an opaque methodology and disregard for industry input and collaboration,” Sarah Gallo, CBA’s senior vice president of product policy, said in an emailed statement.
According to Gallo, the FDA’s proposal “compares recent industry-supported research that has demonstrated that data-driven labels that reinforce important nutrient information, including calories, which nutrients to recommend and which to limit, are most effective in helping consumers eat the healthiest foods.” Not reflected. select.”
Consumer Brands highlighted that many companies are already voluntarily implementing front-of-pack nutrition information using the industry-established Facts up Front label.

Consumer Brands said “hundreds of thousands of food and beverage products” carry the Facts up Front label.
Permission granted by the Consumer Brands Association
Citing member companies, the association said label changes can cost between $1 million and $26 million, depending on the scope of a brand’s product portfolio, and that major label changes in the industry would take three to five years to implement.
Lebor provided the following example from working with a client’s beverage business to implement Facts up Front:His redesign cost over $750,000, with a total company-wide cost of over $4 million.”
“This highlights the financial burden these schemes place on manufacturers in particular, and pose an even greater challenge to small and medium-sized businesses,” she said.
Provided by the U.S. Food and Drug Administration
CSPI has long been critical of Facts up Front. In a statement, Facts up Front said labels “do not indicate whether a product is high in nutrition, have no influence on consumer purchases, and have primarily served to delay this process for more than a decade.” ”
“The next administration has an opportunity to finalize this important issue. Create rules And deliver on your promise to stand up to Big Food,” CSPI wrote. “When finalizing its policy, we hope that the FDA will take into account the growing international evidence supporting the “High In” style labeling adopted by our neighbors, North and South.”
American Beverage said it will continue to work with the Trump administration and the FDA on this nutritional information issue. “That’s why beverage companies pioneered front-of-package labeling more than 10 years ago when we voluntarily added calorie labels to every bottle we sold,” spokesman William Dermody said in a statement.
Redesign prospects
Once the rules are finalized, designers are tasked with developing “a cohesive design that harmonizes with the existing brand.” aesthetics says Ravi Sawhney, founder and CEO of RKS Design, a global packaging design company. CPG.
Consumer testing can inform brands on how to best integrate new labels while capturing consumer attention.
The impact could be different for small and large brands, Sawhney said in an email. Smaller products can handle more limited resources, which means the most popular products may need to be prioritized in an early redesign, he said. Meanwhile, larger companies have better resources to adapt to regulatory changes but face the challenge of ensuring consistency across different product lines.
Companies should consider how more visible labels could impact consumer perception and trust in their brands, Sawhney said. Some people see it as modernizing packaging or rebrand Improve transparency and engage health-conscious consumers.
Michael Keplinger, Director of Testing and Insights at Smashbrand, agreed with the opportunity.
“Given the far-reaching implications for packaging design, all packaging should be redesigned in accordance with these rules. “Brands that approach this strategically must rethink their messaging hierarchy and communication flows to effectively integrate the change,” he said in a written statement. “On the other hand, brands that simply ‘slap in the face’ risk long-term damage to the clarity of their message and consumer engagement.”
The FDA is accepting comments on the proposed rule until May 16. Rainer suspects that key issues in the debate may include the size and prominence of the information box, as well as the thresholds for designating it as low, medium, or high.
“It would be too easy for a product to exceed the 5% threshold for ‘low.’ This is probably not a desirable position for many companies that market their products using an overall packaging narrative that means more. “It’s a healthier product,” she said.
Rainer suspects companies will think about how to achieve tighter, leaner and simpler designs. She said these companies are “the smartest marketers in the world.” “I think they might give some thought to other ways to present this information.”










